🩸 RED BLOOD JOURNAL — TRANSMISSION
T#GENE–TBR // THE BLOOD CODE
Classification: Parasitology / Genome Integrity / Investigative Inquiry
Distribution: Open File
SECTION II — DISSECTING GENE TBR: THE PARASITE’S GENOMIC SHIELD
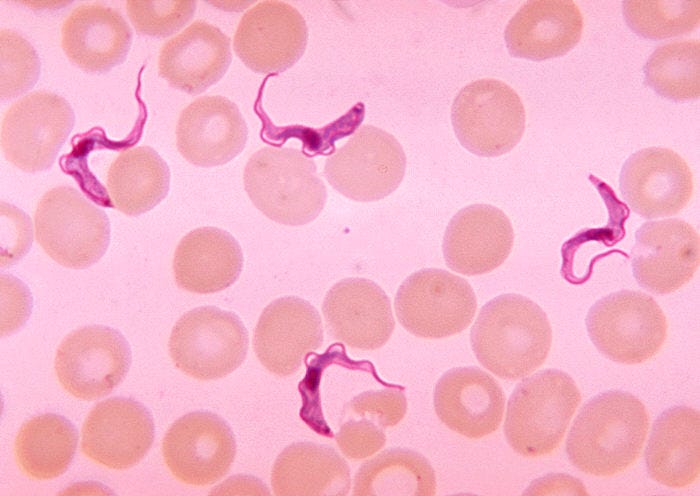
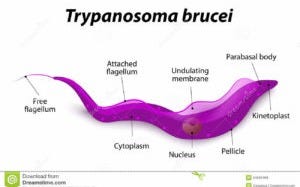
Within the genome of Trypanosoma brucei brucei, researchers have identified genes responsible for maintaining DNA replication under extreme biological stress. Among these is a gene annotated in genomic databases as encoding DNA polymerase κ, an enzyme associated with translesion DNA synthesis (TLS)—a process that allows DNA replication to continue past sites of damage.
This class of polymerase does not repair DNA in the conventional sense. Instead, it enables survival in hostile environments by tolerating genetic lesions that would otherwise stall replication. In blood-borne parasites exposed to oxidative stress, immune attack, and inflammatory signaling, such tolerance is not optional—it is existential.
Genomic mapping places this polymerase within pathways associated with interstrand crosslink repair, grouped in comparative databases under the Fanconi anemia pathway. In humans, disruption of this pathway results in Fanconi anemia, a disorder defined by bone marrow failure, red blood cell depletion, and systemic vulnerability.
The significance is not that the parasite “causes” Fanconi anemia, nor that it reproduces the disease mechanism directly. The significance is architectural:
🧬 A blood-dwelling organism relies on genome-damage tolerance systems whose failure, in humans, manifests as catastrophic blood collapse.
This is not mimicry of disease. It is convergence around survival at the molecular edge—where blood, replication, and failure intersect.
SECTION III — THE BLOOD PARADOX
While T. brucei preserves its own genomic continuity, the host’s blood system degrades.
Trypanosomiasis is consistently associated with progressive anemia, driven by immune activation, inflammatory cytokine signaling, altered erythrocyte turnover, and disrupted red blood cell production. The parasite survives by maintaining genetic stability under stress; the host pays the cost in collapsing hematologic equilibrium.
Two systems. One bloodstream. Opposite outcomes.
This asymmetry is the real anomaly.
SECTION IV — INVESTIGATIVE SIGNALS (FLAGGED)
History records that major sleeping sickness epidemics surged during periods of colonial disruption, forced labor, ecological manipulation, and population control campaigns in sub-Saharan Africa. Official narratives attribute these outbreaks to environmental change and vector dynamics.
Yet the pattern remains:
• sustained exposure
• delayed intervention
• selective urgency
The parasite’s extraordinary genomic resilience—paired with its ability to dismantle host blood systems—raises questions that remain structurally unexplored.
Not accusations.
Unresolved signals.
SECTION V — CONCLUSION: WHERE THE TRANSMISSION BREAKS
Scientifically, Trypanosoma brucei represents one of the most refined examples of blood-borne survival biology known.
Investigatively, it exposes a deeper problem: when organisms that destroy blood are studied only as pathogens—and not as systems—the conversation stops short of power, policy, and intent.
🩸 The parasite adapts.
The host collapses.
The record stays clean.
That is the true “red blood transmission.”
DISCLAIMER
This Transmission integrates established parasitology and genomics with clearly marked investigative inquiry. Speculative elements are presented as hypotheses, not conclusions. This document is not medical advice.
🧬Parasite Gene Mirrors Human Blood Failure.
This investigative report examines the Trypanosoma brucei parasite, which is the biological catalyst for African sleeping sickness.
The author explores how the gene TBR facilitates DNA repair within the parasite, allowing it to survive while simultaneously triggering severe anemia and red blood cell destruction in human hosts.
Beyond conventional science, the text proposes a conspiracy theory suggesting this genetic commonality between the parasite and human blood disorders might indicate bioweapon engineering.
It highlights the parasite's ability to evade the immune system and questions whether colonial history or modern research hides a more sinister origin for the disease.
Ultimately, the source serves as a provocative blend of parasitology and speculative inquiry regarding genetic manipulation.